We need to rethink how we want to tackle MS in 2019. Do we need a true induction approach; i.e. sequential therapies? Or will a simple combination therapy approach suffice?
In 2018 I continued to ask the MS community to ditch the term induction therapy (see ECTRIMS2018 slideshow, and Current Opinion review, below). The term ‘induction’ is not a very useful term when describing the mode of action of alemtuzumab, cladribine and HSCT, because it comes with baggage from other disease areas and is often misrepresented.
A lot of people in the field equate the term ‘induction’ with the use of high-efficacy treatment early on in the course of the disease. For example, many Italian neurologists refer to the use of natalizumab as first-line therapy as an induction strategy. Similarly, the use of mitoxantrone before interferon-beta or glatiramer acetate is referred to as an induction strategy in France (see below). The latter is correct, because induction usually means sequential therapies, with two or three phases of treatment. Induction, followed by a consolidation treatment and finally a maintenance therapy.
I have stopped using the term ‘induction’ outside of the context of mitoxantrone followed by maintenance treatment. I now use the term ‘immune-reconstitution therapy’ or ‘IRT’ to describe short intermittent courses of treatment. This is relevant to alemtuzumab, cladribine, monotherapy mitoxantrone and HSCT.
IRTs have many advantages over maintenance therapies, which I have highlighted in the past and cover in detail in my Current Opinion review. The main advantage is that IRTs (1) tend to be on average highly-effective treatments, (2) they only remain in the body for a short period of time, which is a very useful attribute if you are a woman who is thinking about falling pregnant, (3) they induce long-term remission in some patients, which may turn out to be a cure in the future and (4) they frontload risk. Most of the adverse events associated with IRTs occur early in the treatment hence the term frontloading. In comparison with maintenance therapies, the risks accumulate over time. The latter point is not a trivial point; with immunosuppressive drugs, the risks of opportunistic infections and treatment-related malignancies increases with time.
However, I predict that based on recent insights we will need to use more sequential and combination therapy strategies to treat MS. We are already doing this without our realising it. I reviewed this in two sequential presentations at this year’s ABN in Birmingham.
ABN 2018
However, we are going to need a more systematic approach if we are going to get sequential and combination therapies right. For example, we need more anti-inflammatory in combination with neuroprotection trials. The latter is happening, but too many neuroprotection trials are still going ahead as monotherapy trials.
Another strategy is antivirals in combination with anti-inflammatories. The latter sound left-field, but we need to do these studies to address the EBV-HERV hypothesis. Something we have been pushing is anti-plasma cell agents on top of anti-inflammatories to scrub the CNS clean of OCBs that may be driving progressive MS. We will hopefully start our first trial in this space this year.
The question we have to ask is how ready is the MS community for adopting induction/sequential and combination therapy strategies to manage MS? Based on recent discussions they are not ready enough.
#ThinkCombination #ThinkSequential
CURRENT OPINION REVIEW
Giovannoni G. Disease-modifying treatments for early and advanced multiple sclerosis: a new treatment paradigm. Curr Opin Neurol. 2018 Jun;31(3):233-243.
PURPOSE OF REVIEW: The treatment of multiple sclerosis is evolving rapidly with 11 classes of disease-modifying therapies (DMTs). This article provides an overview of a new classification system for DMTs and treatment paradigm for using these DMTs effectively and safely.
RECENT FINDINGS: A summary of research into the use of more active approaches to early and effective treatment of multiple sclerosis with defined treatment targets of no evident disease activity (NEDA). New insights are discussed that is allowing the field to begin to tackle more advanced multiple sclerosis, including people with multiple sclerosis using wheelchairs. However, the need to modify expectations of what can be achieved in more advanced multiple sclerosis are discussed; in particular, the focus on neuronal systems with reserve capacity, for example, upper limb, bulbar and visual function.
SUMMARY: The review describes a new more active way of managing multiple sclerosis and concludes with a call to action in solving the problem of slow adoption of innovations and the global problem of untreated, or undertreated, multiple sclerosis.
A TRUE INDUCTION THERAPY APPROACH
Edan et al. Mitoxantrone prior to interferon beta-1b in aggressive relapsing multiple sclerosis: a 3-year randomised trial. J Neurol Neurosurg Psychiatry. 2011 Dec;82(12):1344-50.
OBJECTIVES: The long-term impact of interferon-beta-1b (IFN) might be improved by short-term immunosuppression with mitoxantrone (MITOX) in aggressive relapsing-remitting multiple sclerosis (ARMS) patients.
METHODS: In this 3-year clinical and MRI study, 109 ARMS patients (two or more relapses in the previous 12 months and one or more gadolinium (Gd)-enhancing MRI lesion) were randomised into two groups: 54 patients received MITOX monthly (12 mg/m(2); maximum 20 mg) combined with 1 g of methylprednisolone (MP) for 6 months followed by IFN for the last 27 months, and 55 patients received IFN for 3 years combined with 1 g of MP monthly for the first 6 months. The primary endpoint was the time to worsen by at least one Expanded Disability Status Scale point confirmed at 3 months.
RESULTS: The time to worsen by at least one Expanded Disability Status Scale point confirmed at 3 months was delayed by 18 months in the MITOX group compared with the IFN group (p<0.012). The 3-year risk of worsening disability was reduced by 65% in the MITOX group relative to the IFN group (11.8% vs 33.6%). MITOX patients had a reduced relapse rate by 61.7%, a reduced number of Gd-enhancing lesions at month 9 and a slower accumulation of new T2 lesions at each time point.
CONCLUSIONS: Although there were limitations in this investigator-academic-driven study, the data do suggest that mitoxantrone induction therapy prior to INF beta-1b may have a role in aggressive disease.
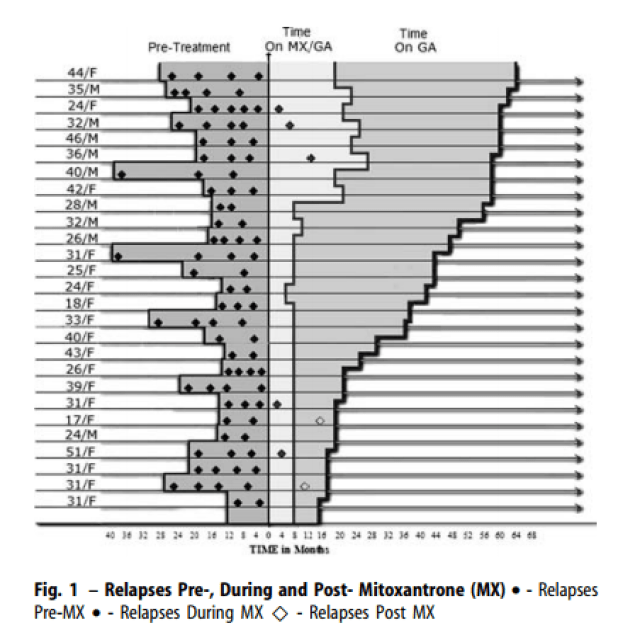
Ramtahal et al. Sequential maintenance treatment with glatiramer acetate after mitoxantrone is safe and can limit exposure to immunosuppression in very active, relapsing remitting multiple sclerosis. J Neurol. 2006 Sep;253(9):1160-4. Epub 2006 Sep 21.
Background: Mitoxantrone has been approved by the FDA for worsening relapsing remitting and secondary progressive Multiple Sclerosis. However the benefits of this agent in reducing disease progression and relapse rate cannot be sustained in the long-term, as treatment is limited by the potential for cumulative cardiotoxicity.
Objectives and methods: We report our experience utilising Glatiramer Acetate as maintenance immuno-modulatory treatment following initial immunosuppression with Mitoxantrone in a consecutive series of 27 patients with very active relapsing remitting disease, eight of whom had experienced continuing relapse activity on first-line treatment.
Results: Duration of treatment with Mitoxantrone and thereby cumulative dose were reduced as our experience with the combination increased.No unanticipated side effects of combination treatment were encountered over a follow-up period of 66 months. A single patient developed therapy related acute leukaemia (TRAL) 9 months after completion of Mitoxantrone.A sustained 90% reduction in annualised relapse rate (p < 0.001) has been observed. Disability is stable or improved in all patients a mean of 36 (16-66) months from initiation of treatment. Early suppression of relapse activity with Mitoxantrone has been maintained at a mean of 22 months from last dose of this agent. Only two relapses have occurred in the cohort since withdrawal of Mitoxantrone, occurring in the two patients who had previously been treated with Glatiramer Acetate. In 9 of the first 10 patients treated, imaged a mean of 27 months after withdrawal of Mitoxantrone, no enhancing lesions were identified on MRI brain scans.
Conclusions: Glatiramer Acetate appears a safe and effective option for continuing disease modification in patients with relapsing remitting multiple sclerosis treated with Mitoxantrone. The treatment protocol utilised in later patients in this series appears to have the potential to limit exposure to this agent.