In response to a small spat that happened amongst two commentators yesterday, I want to stress that the Barts-MS blog is not a general purpose information resource for people with MS. Our brief is to ‘interpret good, bad and other research news’. We do provide information about how we at Barts-MS manage MS, but please note this information may be at odds with advice you have been given, your own MS centre’s practice, or advice from other healthcare professionals. You have to realise that the information provided by the MS charities has to be ‘evidence-based’ and if not evidence-based derived by ‘consensus’. Consensus refers to ‘generally accepted opinion or decision among a group of people’. If you want a consensus opinion this blog is not the place you should come to for guidance. Consensus is typically reached when adoption of a process has reached the late majority, in other words it is behind the peak of the curve.
Case Study:
Last week I saw a patient with PPMS (>55 years of age). He has had symptomatic disease ~15 years and is using a stick for outdoor mobility. He has a dropped left foot that gets worse when he is tired and as a result has had several falls. Fortunately, he has had no fractures. Other problems included sexual dysfunction, urinary hesitancy, urgency, urgency incontinence and nocturia, constipation, back pain, restless legs and nocturnal leg spasms, poor sleep, fatigue, low mood and the ubiquitous anxiety about the future. He has previously been part of the fingolimod in PPMS study, but doesn’t feel fingolimod helped. His walking had continue to deteriorate. His latest MRI showed several spinal cord, cerebellar, pontine and brain lesions. There was gross spinal cord and cerebral atrophy. None of the lesions enhanced after the administration of gadolinium (contrast agent), indicating that none of the lesions were active. His recent cerebrospinal fluid analysis showed OCBs and a raised CSF neurofilament light chain level (1050 ng/ml; ~ double the upper limit of normal for his age). What should I do for this patient?
He has been following our blog for years and wanted something active done about his MS. He had read the literature and was keen to have something done about his disease progression. He was dissatisfied with his current neurologist’s attitude who had said there is ‘nothing to be done about your disease at present’. He has taken things into his own hands and had ordered high-dose biotin from a US-based compounding pharmacist, but has not noticed a response. After reading around he is keen to go onto low dose methotrexate; his was particularly concerned about losing arm and hand function and is convinced that our group are right about our length-dependent axonopathy hypothesis.
Should I listen to him and prescribe him off-label methotrexate or not? Should I say no and ask him to wait for ocrelizumab? Please note there is no guarantee that ocrelizumab will be licensed by the EMA for PPMS and not guarantee that it will be green-lighted by NICE. In addition, he is over 55, has an EDSS of 6.0 and no active lesions on MRI; this may be important as I suspect the EMA/NICE will want to define a subgroup of people with PPMS who are more likely to respond to ocrelizumab (for example, young (less than 50/55), less disabled (EDSS <=5.5), with active, or Gd-enhancing, lesions on MRI). Should I put him on a waiting list for upcoming clinical trials?
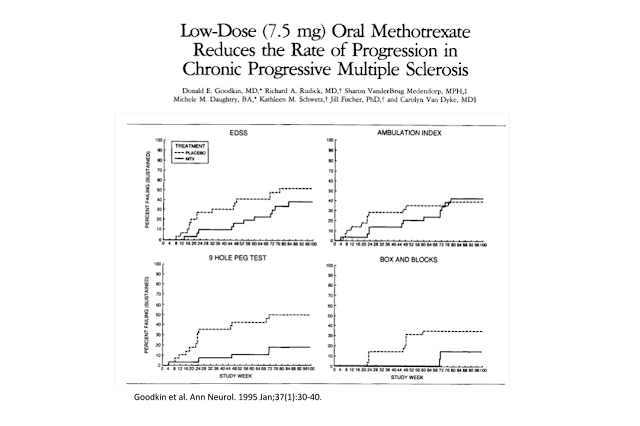
Introduction and Methods: A randomized, double-blinded, placebo-controlled, clinical trial of low-dose, weekly, oral methotrexate was performed in 60 patients with clinically definite chronic progressive multiple sclerosis (MS) attending a referral-based outpatient MS clinic. Study patients were 21 to 60 years old with a disease duration of longer than 1 year. Patients’ Expanded Disability Status Scale scores were 3.0 to 6.5 (ambulatory with moderate disability). Patients were first stratified by Expanded Disability Status Scale scores, 3.0 to 5.5 and 6.0 to 6.5, and then were randomized to receive methotrexate or placebo treatment. Treatment consisted of weekly, oral, low-dose (7.5 mg) methotrexate or identical placebo for 2 years, followed by observation for as long as 1 year.
Results: A composite outcome measurement instrument was used and consisted of (1) Expanded Disability Status Scale, (2) ambulation index, (3) Box and Block Test, and (4) 9-Hole Peg Test. Failure of therapy was indicated by a designated change that was sustained for more than 2 months in one or more components of this composite measure. Significantly less progression of impairment as measured by validated tests of upper-extremity function was observed in the methotrexate treatment group in the absence of clinically significant toxicity.
Conclusions: We conclude that low-dose, weekly, oral methotrexate offers a new, relatively nontoxic treatment option for patients with chronic progressive MS.
CoI: multiple

I only come to this site because it is ahead of the curve. By reading the posts on this site I know more than my neurologist about what is going on in the field.
So ProfG, the current consensus view is watch and wait but is that your opinion? Or, are you going to offer him cladribine (assuming the gent hasn't already precluded that himself)? He seems a perfect fit for the Barts off-label clad programme and in his position, I would go for it.
Good call!
If I may say so – I find that the UK MS Trust take _all_ evidence into consideration, also the more tenuous, cutting edge stuff. They certainly did in their answers to my questions. But – and this is vital – they also include side-effect information and respect the fact that not everyone wishes to go for high risk treatment or treatment which may or may not work. They are cutting edge, but well-rounded.I cannot praise them highly enough. What they say on their car stickers is certainly true: "Making sure no one has to deal with MS alone."I know I'm preaching to the converted here, but they have taken massive burdens of bewilderment and anxiety away from me. They are indespensible and wonderful.
The problem I have with your article is that it presumes that all eventually adopted treatments are effective. In many cases, the "laggards" are actually those who are refusing to prescribe ineffective unproven treatments. The history of medicine is filled with commonly used but ineffective treatments (cardiac catheterization for stable angina for example). In progressive MS treatment, we are particularly guilty of prescribing treatments which have no evidence or are proven to be ineffective or marginally effective. As you describe yourself, there is a good chance that ocrelizumab is ineffective in older patients with long-standing non-relapsing progressive multiple sclerosis without MRI activity. Should we prescribe medications which are very expensive and have significant safety concerns just to appease our psychological need to "do something" or "be aggressive?" It's all fun and games until someone gets a side effect or we deplete our limited resources so that we are unable to provide proven effective therapies to our patients. The next time you want to prescribe natalizumab to patient with early active RRMS and get denied by the NHS, who are you going to complain to? Just because you are not taking a well-marketed pharmaceutical does not mean you are "doing nothing." Some alternative treatments could include high dose biotin, exercises regimens, nutrient dense diets, and symptomatic pharmaceutical treatments. For the sake of the integrity of our profession, I think it is very important that we be honest and straight-forward about what we can and what we cannot achieve. Our treatment of chronic progressive multiple sclerosis is currently lacking, and we are in need of a revolution.
I would normally simply agree with what you have written here, as a pwppms. I say to myself quite a lot – just be at peace, there is nothing to be done, look the truth in the eye. And I get on with loving life and being "brave", both for myself and my loved ones.But in the last few days, I have had more problems with my hand and arm function. This is really frightening, makes me feel vulnerable and terrified of losing independence. So frightening is this, that I caught myself "taking comfort" in the following: …I did have the experience recently, when very fatigued, of finding it difficult to cough properly. Perhaps my breathing muscles are slightly affected. And maybe, if things do get really bad, this element will worsen also, and I will just stop breathing in my sleep and slip away quietly, before I become too dependent on others…Then I told myself to pull myself together and stop being such a coward. And I thought of the excellent "Think Hand" campaign, which may result in something being found and proven to really help people like me. And no longer will we be going to neurologists who all too readily dismiss us as hopeless cases, or a waste of resources.
Are there any other trials showing the same effect of MTX? Because if there are, why isn't cheap and cheerful MTX used in progressive MS? Or was this a fluke study that nobody managed to replicate?
I keep thinking about this poor man and wondering if he really is a patient and is following every comment or is he merely hypothetical? Whichever, if you believe cladribine better than LDM and appropriate then that is what you should offer. I don't understand the significance of high number neurofilaments in CSF (other than they shouldn't be there), does this imply damage is recent/ongoing and therefore DMT will help? Or do filaments shed as axons die, maybe long after demyelination? He also has OCBs in CSF, if OCBs indicate IGs present in CSF, does that not suggest active disease? Sorry if I'm being thick, am only a pwMS…If you start this patient on cladribine, could he potentially also join PROXIMUS trial after six months? He sounds keen to try anything, even if he is hypothetical 😉
high levels of neurofilaments means nerve damage is present, it may mean current DMT can help but also could be that progression is ongoing that may not optimally respond to DMT
I come to the blog to really inform me, keep up to date on what Science knows so far about what I have, because even as I do treatment through SUS (NHS here in Brazil) I have a medication protocol to follow, so unless I am "willing" to pay and find a neurologist "willing" to prescribe a medicate outside of the SUS protocol has no way to me to medicate"radically". But talking about the case exposed: why not risk medicating he with Cladribine, or maybe a Alemtuzumab, but Cladribine would be a good option. He doesn't have time to wait for Ocrelizumabe to be licensed.I come to the blog to really inform me, keep up to date on what Science knows so far about what I have, because even as I do treatment through SUS I have a medication protocol to follow, so unless I am " Willing "to pay and find a neurologist" willing "to prescribe a drug outside the protocol of the SUS has no way to self medicate" radically. " But talking about the case exposed: why not risk medicating it with Cladribine, or maybe a Alemtuzumabe, but Cladribine would be a good option. He does not have time to wait for Ocrelizumabe to be licensed. I know some people with NMO who use Metotrexate, for some the relapses secure, for others not.
Its a no brainier (no pun intended :-)). Do what you can to help the man. The patient sounds informed. Doing nothing will see the man go downhill for sure. Another life devastated by the disease. I have to smile at the term "watchful waiting". What does that look like exactly? From what I read this seems to equate to brief yearly consultations (if lucky).
Re: "watchful waiting"You watch, assess, wait, watch, assess, wait and keep doing this until a licensed treatment emerges for treating PPMS. Some called this 'best supportive care'.